 Removing one electron from the orbital still leaves one unpaired electron in the other orbital. The electron would be removed from the orbital, as this is the highest in energy. Therefore, Bond order = 1 / 2 Īs a result, the bond order of = 1 / 2 = 1 / 2 (4)=2 is 2.įor O 2 + molecule, an electron is removed from * 2 p y orbital. Explanation: We can work this out by looking at the molecular orbital diagram of O2 O+ 2 has 1 fewer electron than O2 which is what gives it the positive charge. However, this picture is at odds with the magnetic behavior of oxygen. There is an OO double bond, and each oxygen atom has eight electrons around it. 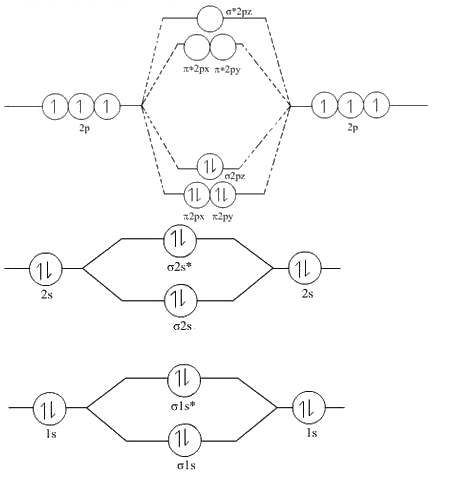
There are 10 bonding and 6 nonbonding electrons in the orbitals according to the molecular orbital configuration.Īccording to the molecular orbital configuration, there are 10 bonding and 6 nonbonding electrons in the orbitals. We would write the following Lewis structure for O 2: This electronic structure adheres to all the rules governing Lewis theory. The molecule O 2 has the following molecular orbital configuration: A total of 16 electrons make up an O 2 molecule. In MO theory, a star (*) sign always indicates an anti-bonding orbital.įollowing the aufbau ('building up') principle, we place the two electrons in the H 2 molecule in the lowest energy molecular orbital, which is the (bonding) sigma orbital.To begin, we can write the molecule’s molecular orbital arrangement. The second, sigma-star ( σ *) orbital is higher in energy than the two atomic 1 s orbitals, and is referred to as an anti-bonding molecular orbital. According to MO theory, the first sigma orbital is lower in energy than either of the two isolated atomic 1 s orbitals – thus this sigma orbital is referred to as a bonding molecular orbital. When two atomic 1 s orbitals combine in the formation of H 2, the result is two molecular orbitals called sigma ( σ) orbitals. The bonding in H 2, then, is due to the formation of a new molecular orbital (MO), in which a pair of electrons is delocalized around two hydrogen nuclei.Īn important principle of quantum mechanical theory is that when orbitals combine, the number of orbitals before the combination takes place must equal the number of new orbitals that result – orbitals don’t just disappear! We saw this previously when we discussed hybrid orbitals: one s and three p orbitals make four sp 3 hybrids. These two new orbitals, instead of describing the likely location of an electron around a single nucleus, describe the location of an electron pair around two or more nuclei. Oxygen atom has 2s and 2p valence orbitals and 6 valence electrons: Each oxygen contributes 6, so we distribute 12 valence electrons into the molecule to get O2. In molecular orbital theory, we make a further statement: we say that the two atomic 1 s orbitals don’t just overlap, they actually combine to form two completely new orbitals. You'll need the molecular orbital (MO) diagram of O2. We can ignore the 1s orbitals, because they do not contain the valence electrons. 
Then we rank them in order of increasing energy. 
When we described the hydrogen molecule using valence bond theory, we said that the two 1 s orbitals from each atom overlap, allowing the two electrons to be shared and thus forming a covalent bond. Before we can draw a molecular orbital diagram for B, we must find the in-phase and out-of-phase overlap combinations for borons atomic orbitals. Let’s consider again the simplest possible covalent bond: the one in molecular hydrogen (H 2). \)Īnother look at the H 2 molecule: bonding and anti-bonding sigma molecular orbitals Oxygen needs to bond twice, shown as the lone dots on the left and right sides of the oxygen atoms in the below diagram. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
